Minerva Surgical, Inc. Third Quarter 2021 Financial Results
IR News
Click here for PDF Version
December 2, 2021
SANTA CLARA, Calif., Dec. 02, 2021 (GLOBE NEWSWIRE) -- Minerva Surgical, Inc. (Nasdaq: UTRS) (Minerva Surgical or the Company), a medical device company that develops therapeutic devices that treat abnormal uterine bleeding in a minimally invasive manner, today reported financial results for the third quarter ended September 30, 2021 and provided full-year 2021 revenue guidance.
Recent Business Highlights
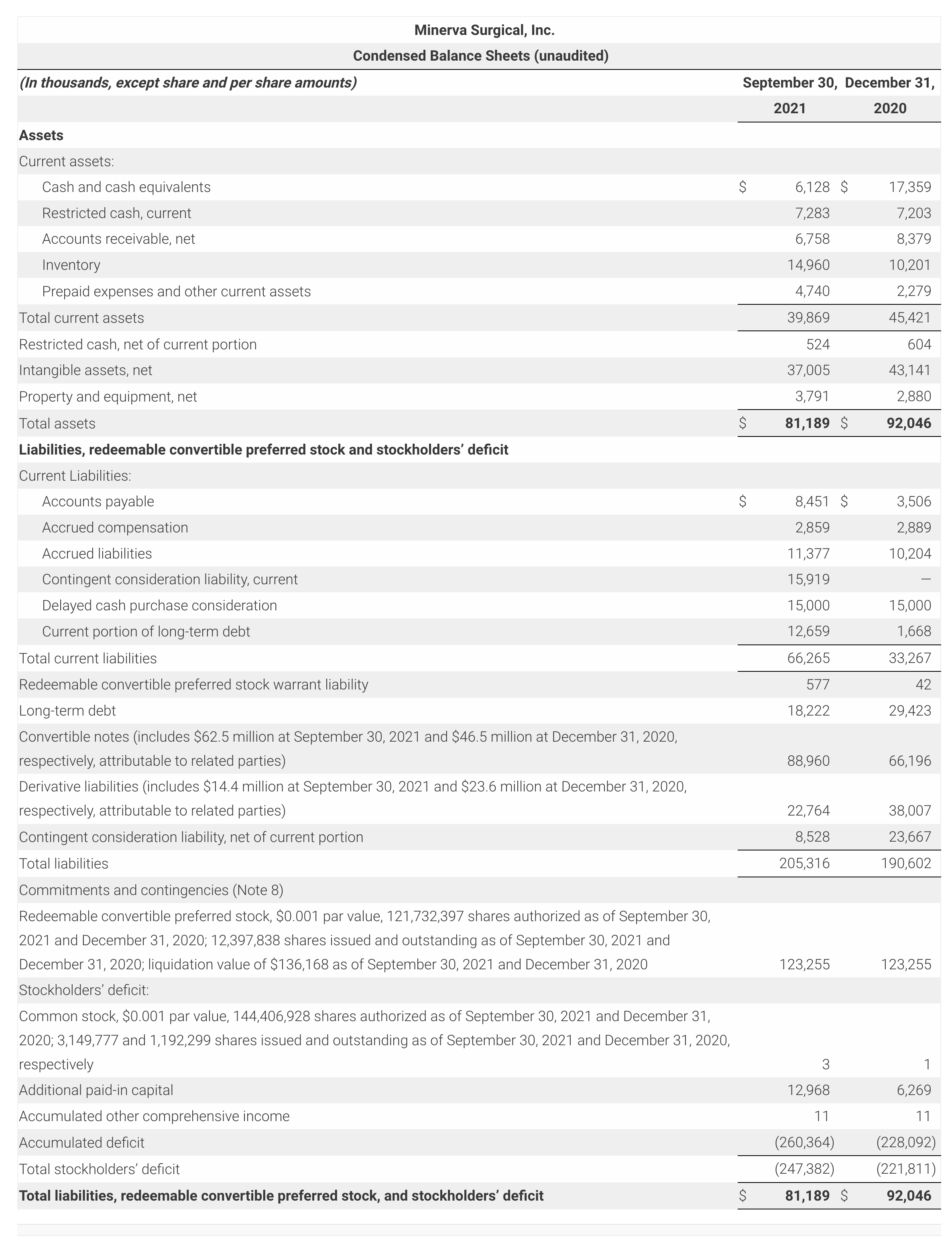
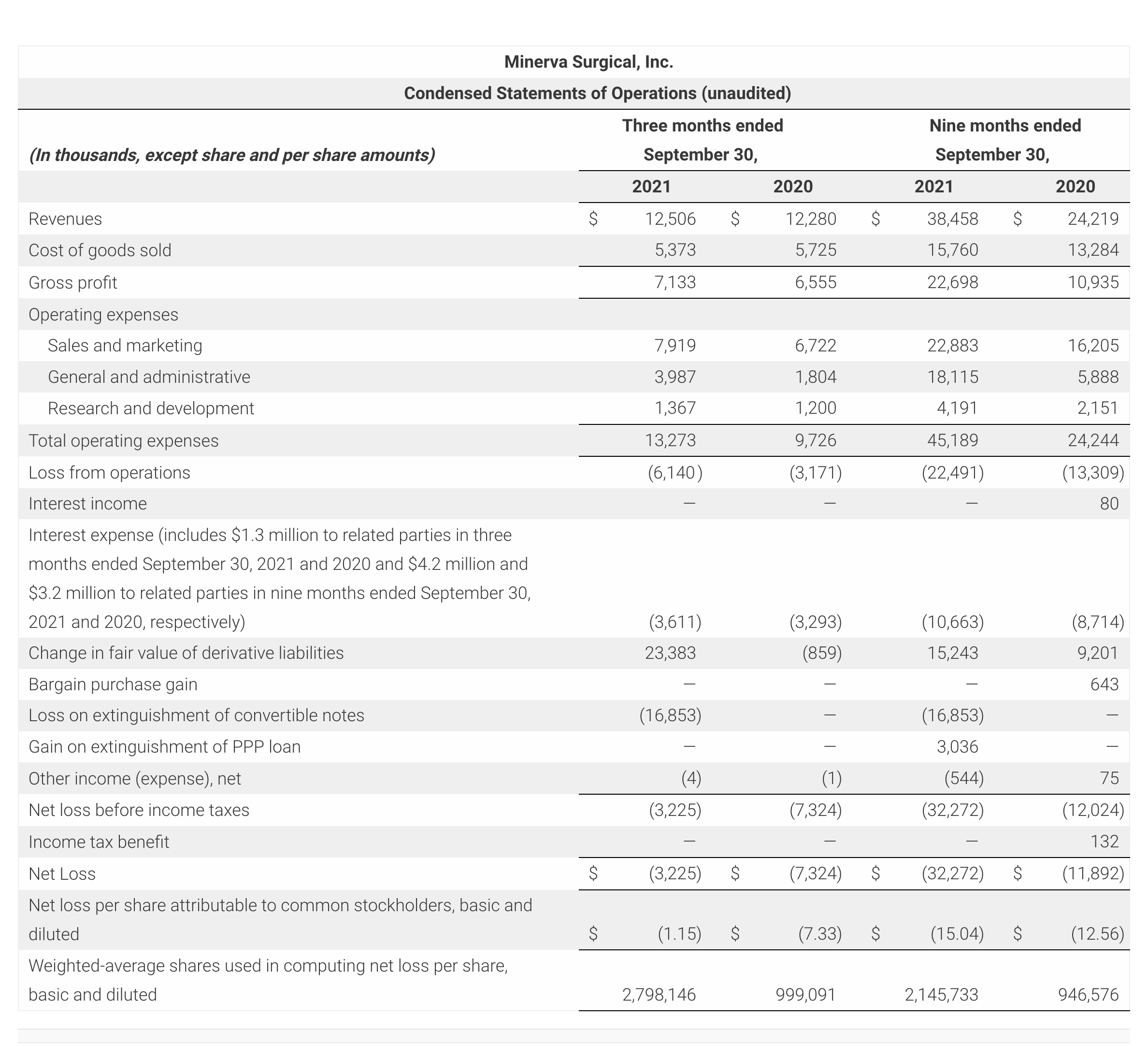
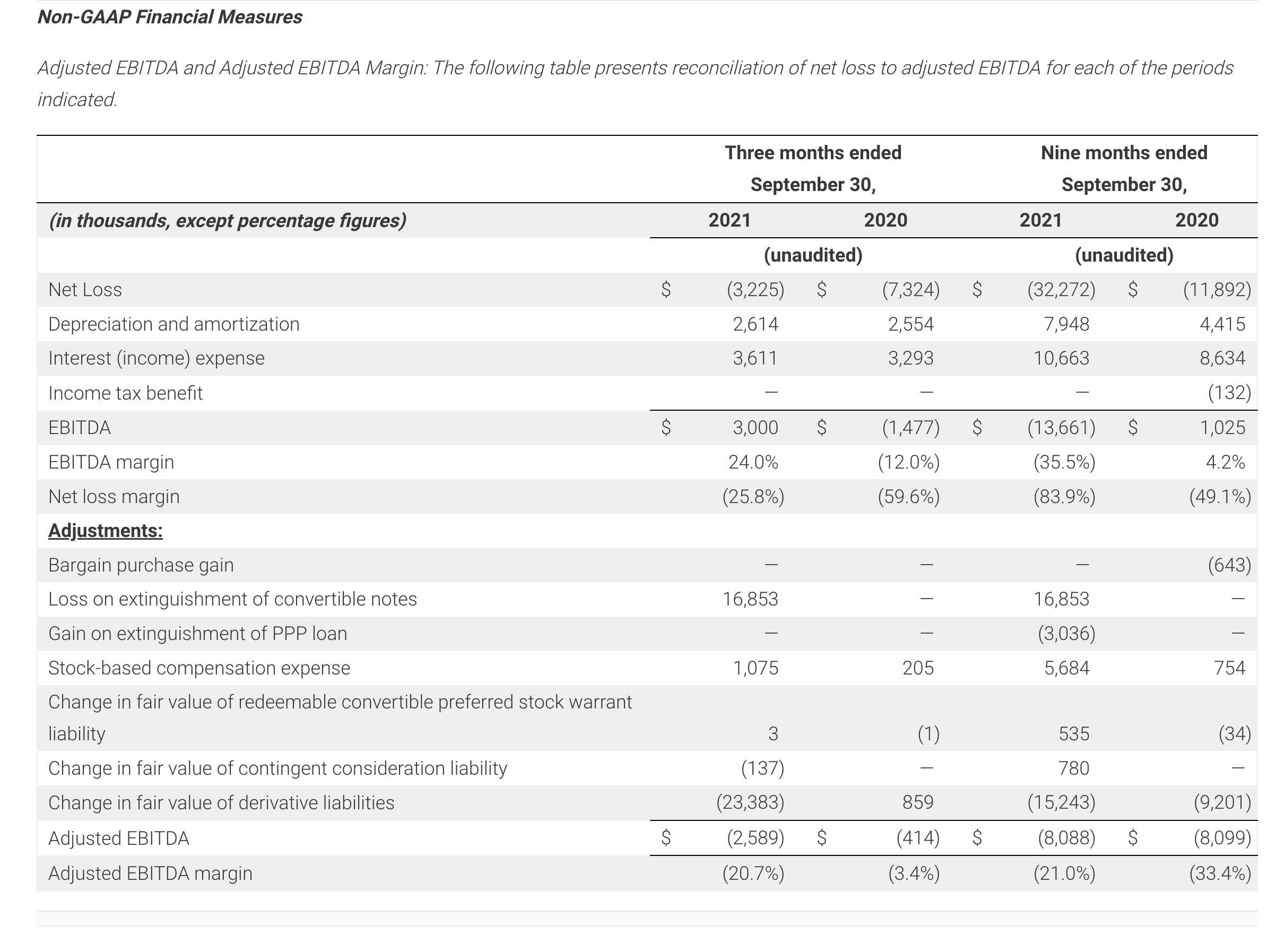
- Reported third quarter 2021 revenues of $12.5M, compared with revenues of $12.3M in third quarter of 2020.
- Reported revenue of $38.5 million for the nine-month period ended September 30, 2021 representing a 58.8% increase compared to $24.2 million for the same period in 2020.
- Successfully closed the Company’s initial public offering on October 26, 2021, with net cash proceeds to the Company of $69.8M.
- In October 2021, refinanced its remaining outstanding debt with a new $40 million term loan facility, substantially reducing its net interest costs over the next five years.
- Continued the expansion of the U.S. sales team to accelerate growth in 2022.
- Launched AUBandME.com, a patient educational website for the estimated 18 million women in the U.S. suffering from heavy menstrual bleeding.



